Article Text
Abstract
Introduction 2-Chloroacetophenone (CN), o-chlorobenzylidene malonitrile (CS) and oleoresin capsicum (OC) are common riot control agents. While serious systemic effects are uncommon, exposure to high concentrations may lead to severe complications and even death. The aim of this narrative review is to summarise all main aspects of the riot control agents CN, CS and OC toxicology, including mechanisms of toxicity, clinical features and management.
Methods OVID MEDLINE and ISI Web of Science were searched for terms associated with CN, CS and OC toxicity in humans and those describing the mechanism of action, clinical features and treatment protocols.
Results CN, CS and OC are effective lacrimating agents; evidence for toxicity, as measured by the threshold for irritation, is greatest for CN, followed by CS and OC. Typically, ocular and respiratory tract irritation occurs within 20–60 s of exposure. Ocular effects involve blepharospasm, photophobia, conjunctivitis and periorbital oedema. Following inhalation, effects may include a stinging or burning sensation in the nose, tight chest, sore throat, coughing, dyspnoea and difficulty breathing. Dermal outcomes are variable, more severe for CN and include dermal irritation, bulla formation and subcutaneous oedema. Removal from the contaminated area and fresh air is a priority. There is no antidote; treatment consists of thorough decontamination and symptom-directed supportive care. Ocular exposure requires thorough eye decontamination, an eye exam and appropriate pain management. Monitoring and support of respiratory function is important in patients with significant respiratory symptoms. Standard treatment protocols may be required with patients with pre-existing respiratory conditions. Dermal exposures may require systemic steroids for patients who develop delayed contact dermatitis.
Conclusions CN, CS and OC are effective riot control agents. In the majority of exposures, significant clinical effects are not anticipated. The irritant effects can be minimised both by rapid evacuation from sites of exposure, decontamination and appropriate supportive care.
- 2-Chloroacetophenone; o-Chlorobenzylidenemalonitrile; CS; CN; Oleoresin capsicum OC; pepper spray
- medical review
Statistics from Altmetric.com
- 2-Chloroacetophenone; o-Chlorobenzylidenemalonitrile; CS; CN; Oleoresin capsicum OC; pepper spray
- medical review
Introduction
The chemical agents 2-chloroacetophenone (CN; Mace), o-chlorobenzylidene malonitrile (CS) and oleoresin capsicum (OC; pepper spray) are highly potent irritant incapacitating agents commonly used by law enforcement agencies as a non-lethal option for subduing combative and violent suspects, for crowd control purposes in times of civil disorder and for alleviating siege and hostage situations.1–3 Military organisations have also used these agents for similar purposes, for training4 (Figure 1) and as chemical warfare agents. The US military used CS during the Vietnam War for tunnel denial and crowd control.1 ,5 ,6 Since the Entry Into Force of the Chemical Weapons Convention in 1997,7 these agents have been banned as a method of warfare. However, under a 1975 presidential order, the US military can still use these agents in war zones under limited defensive circumstances with the approval of top military commanders, for example, for controlling rioting prisoners.8
US Navy Seabees in full mission-oriented protective posture gear training with smoke grenades and tear gas. Public domain image. Photograph credit: Photo by Chief Mass Communication Specialist Michael B. Watkins, US Navy.
CN, also known as Mace, was initially the most widely used agent by law enforcement agencies; however, in more recent years, CS has largely replaced CN.9 Products containing CN and CS are also available as a handheld spray for personal self-defence and protection. OC pepper spray has over recent years become increasingly popular with law enforcement agencies, having replaced both CN and CS for civilian use. These agents are commonly referred to as tear gases, riot control agents, harassing agents, incapacitating agents and lacrimators. They are typically dispersed via aerosol, and following exposure they cause immediate and intense eye, nose, mouth, skin and respiratory tract irritation that can lead to temporary incapacitation of exposed individuals. While serious systemic effects are uncommon, exposure to high concentrations may lead to more serious complications and even death. An understanding of toxic effects produced and medical treatment required following exposure is therefore essential for successful first-line and ongoing medical management. This article reviews the three most commonly used chemical riot control agents—CN, CS and OC—and presents an up-to-date overview of the mechanism of action, symptoms expected and medical management required following exposure.
Methods
A literature review was performed by searching OVID MEDLINE (January 1950–January 2013) and ISI Web of Science (1900–January 2013). Bibliographies of articles were screened for additional relevant studies including non-indexed reports. This review identified 229 papers, excluding duplicates. This list was screened for those associated with CN, CS and OC toxicity in humans and those that concisely described the mechanism of action, clinical features and treatment protocols. Articles employed in this review included case reports, case series, animal studies and review articles that were considered relevant. Ninety-six articles were considered relevant.
Chemical properties
CN (CAS 532-27-4) has a molecular formula of C8H7ClO and a molecular weight of 154.59 (Figure 2).10 It has a melting point of about 58–59°C, a boiling point of 244–245°C, and at 20°C it has a low vapour pressure of 5.4×10–3 mm Hg.10 CN is practically insoluble in water, though it is freely soluble in ethanol, ether and benzene. CN was developed at the end of the First World War, although it was not used during combat.11 Following the First World War, it was widely used both by the military and various law enforcement agencies until the development of the more potent and less toxic incapacitant, CS, which became available in 1959.1 Table 1 details a brief comparison of the toxicity of all three agents.
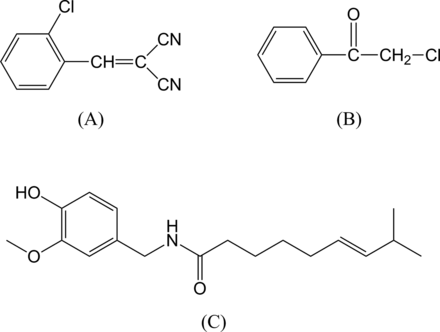
Chemical structures of o-chlorobenzylidene malonitrile (A), 2-chloroacetophenone (B) and capsaicin (C).
CS (CAS 2698-41-1) has a molecular formula of C10H5ClN2 and a molecular weight of 188.6; it was discovered in 1928 by the British chemists Ben Corson and Roger Stoughton, the common name CS being derived from the first letter of their two surnames.9 ,12 It has a cyanocarbon structure (Figure 2). It is a white crystalline solid with pepper-like odour with a melting point of about 93°C and a boiling point of 310°C. It has a low vapour pressure, is sparingly soluble in water while being soluble in acetone, methylene chloride, ethyl acetate and benzene. It hydrolyses somewhat slowly in water, producing o-chlorobenzaldehyde and malononitrile.9 ,10
OC, capsaicin (N-(4-hydroxy-3-methoxybenzyl)-8-methylnon-trans-6-enamide, CAS 8023-77-6), present naturally in the capsicum group of herbs and shrubs, has a molecular formula of C18H27NO3 and a molecular weight of 305.41 (Figure 2).10 It is an odourless, pungent tasting off-white solid with a melting point of about 65°C and a boiling point of 210–220°C.10 ,13 It has a low vapour pressure, is practically insoluble in water while being freely soluble in alcohol, ether, chloroform and benzene.10
Mode of application
Due to these chemical properties, these agents are typically dispersed as fine powdered particles, for example, as a smoke by means of pyrotechnic mixture, via fogging machines or as vapour generated from pressurised liquid systems (aerosolisation). In the training scenario, pyrotechnic pellets produce a fine particulate aerosol. In the medical literature, the terms spray and aerosol tend to be used interchangeably. However, there are distinctions between the different forms of lacrimators. Aerosol typically refers to products that are airborne dispersions used to affect many people in an area such as crowd control, whereas sprays are typically handheld canisters containing the active agent in solution; this is sprayed directly at a single person to incapacitate them. Occasionally liquid products may be dispersed directly at a larger number of people by being added to water and dispersed by water cannon or similar larger-scale devices.14–16 The devices typically used to disperse these agents include bombs, large spray tanks, grenades or canisters that can either be thrown or shot as projectiles, or smaller, handheld spray devices.1 ,9 ,14
Mechanisms of toxicity
The mechanism of action of these agents in humans is not fully understood. Both CS and CN are thought to act as SN2-alkylating agents, reacting readily with nucleophilic sites.1 Important targets include thiol and sulfhydryl groups of enzymes, including lactic dehydrogenase, glutamic dehydrogenase and pyruvic decarboxylase. Inactivation of these metabolic enzyme systems may be related to tissue injury that occurs following exposure.1 ,14 ,17 ,18 Transient receptor potential (TRP) channels are present in airway sensory fibres, with TRPV1 (vanilloid) receptors found to be sensitive to OC. The result is a sensation of pain, but also an inflammatory response, due to the process of neurogenic inflammation.13 This involves release of neuropeptides (including importantly substance P) at the terminals of the C (and special A) fibres, both peripherally and at their junction in the dorsal horn of the spinal cord. These neuropeptides in turn provoke inflammation.
Another subtype, TRPA1, was subsequently shown to be the sensory neuronal receptor for mustard oil (allyl isothiocyanate) and widely sensitive to other reactive irritants, including CS. Both TRPA1 and V1 receptors are thought to be the final common pathway for inflammatory signalling pathways.19 An additional mechanism of action for the irritant and painful effects may be due to bradykinin release.20 CS also contains two cyanogenic groups (Figure 2), and while they may contribute to the local irritant effects of the compound, under normal circumstances it is not thought that enough cyanide would be liberated to cause systemic effects. However, experimentally minimal amounts of cyanide and thiocyanate can appear in the urine following intravenous or oral administration of CS.9 ,18 ,21
Clinical features
The effects of these agents are related to the concentration of the compound and duration of exposure. High concentrations over short periods may be more hazardous than the same dose delivered as low concentrations over longer time periods.20 Evidence for toxicity, as measured by the threshold for irritation (Table 1), is greatest for CN (1.0 mg m3), followed by CS (0.004 mg m3) and OC (0.002 mg m3). In contrast, OC and CS are less lethally toxic than CN (Table 1). Nevertheless, deaths have been reported following exposure to CN, typically at high concentrations in an enclosed space for an extended period of time.22–24 While some concerns have been raised about the safety of CS and OC,25 they are regarded as generally safe when used appropriately.9 ,18 ,26
The eyes and respiratory systems are the primary target organs, with onset of ocular and respiratory tract irritation occurring within 20–60 s.3 The ocular symptoms include pain, blepharospasm, photophobia, conjunctivitis, diffuse conjunctival and scleral injection, periorbital oedema, eyelid erythema and lacrimation.27–47 They do not typically cause irreversible eye effects, but more severe ocular injuries have been reported, including hyphema, uveitis, necrotising keratitis, coagulative necrosis, symblepharon, secondary glaucoma, cataracts and traumatic optic neuropathy and loss of sight.48–52 However, these injuries were noted following exposure to explosive devices discharged near the face and eyes. Additionally, some reports suggest that the carrier solvent used with some sprays may contribute to corneal abrasions.53 ,54 This makes it difficult to determine if the ocular damage was due to the tear gas itself, the carrier solvent or as a result of the explosive discharge of the product.17
Following inhalation, effects can include a stinging or burning sensation in the nose, tightness and pain in the chest, sore throat, sporadic breath holding, dyspnoea, coughing, sneezing and difficulty breathing.27–33 ,36 ,38 ,40 ,42 ,43 ,55–58 Copious rhinorrhoea may occur along with salivation and burning sensation in the mouth and tongue.24 ,28 ,39 ,56 Contaminated saliva that is swallowed may lead to epigastric discomfort and may contribute to nausea and vomiting and/or diarrhoea.27 ,28 ,32 ,39 Persistent coughing may also contribute to retching.
Panic and agitation are common, especially on a first exposure. Other less specific symptoms may include headache, fever, syncope, dizziness and tachycardia.27 ,28 ,31 ,32 ,36 ,39 ,55 ,59 Further systemic effects are unlikely to occur from typical exposures; however, if used in very high concentrations or within confined non-ventilated areas, more severe effects are possible, including bronchospasm, laryngospasm, haemoptysis, reactive airways dysfunction, chemical pneumonitis, pulmonary oedema, asphyxia, heart failure, hepatocellular damage and death.4 ,22 ,24 ,37 ,38 ,56 ,60–63 Some subjects may develop hypersensitivity reactions with fever and pulmonary involvement.60 ,64
Dermal contact to CS typically causes a tingling or burning sensation,16 ,28 ,34 ,37 ,39 ,40 ,43 ,65 ,66 but prolonged exposure can lead to a range of other adverse effects, including erythema, oedema, blistering and superficial burns.16 ,30–32 ,36 ,41 ,43 ,56 ,59 ,65 ,67–71 The latter more severe dermal symptoms are more commonly encountered with exposure to CN, which tends to cause more severe dermal injuries than CS. Aerosol and liquid contact with CN typically leads to pruritis, localised pain, erythema, rash, purpura, desquamation, vesicles, blistering, second-degree burns, bulla, scaling and subcutaneous oedema.45 ,55 ,59 ,72–78 Topical OC products used as pain treatments can also cause burning, stinging and erythema,79 and have also caused contact dermatitis following exposure during food preparation.80
Severe dermal manifestations appear more common where handheld spray products are used. The solvents in these products may contribute to the cutaneous complications. Additionally, adverse skin effects may be accentuated by moisture such as from perspiration, lacrimation, rhinorrhoea or high humidity.17 ,27 ,28 ,32 ,67 ,71 Further dermal effects, including irritant or allergic contact dermatitis and acute generalised exanthematous pustulosis, may be delayed in onset, not presenting until 12–24 h or longer post-exposure.41 ,43 ,59 ,65 ,69 ,70 ,72–75 ,77 ,78 ,81 ,82 Additional risks in riot control situations where tear gases are used include burns from direct contact with the hot metal canister used to disperse these agents or burns caused by the flame generated by the pyrotechnic mixture used.31 ,68 ,82
The unpleasant effects produced tend to force those exposed to seek fresh air,9 and the majority of irritant effects typically resolve within 10–30 min if patients are quickly removed from the source.3 ,5 ,18 ,27 ,28 ,30 However, some effects, especially respiratory effects such as cough and impaired respiratory function, may persist for longer periods in some situations.27 ,30 ,32 ,33 ,38 ,43 ,56 Visual acuity typically returns to normal rapidly while erythema of the lid margins and photophobia may persist longer.27 Rhinorrhoea and salivation may persist for 12 h, and headaches can be prolonged for up to 24 h.27 ,28 Dermal erythema generally subsides within 45–60 min while effects such as blistering and irritant contact dermatitis typically heal with drying of the blistered area within 4 days and minimal scarring after 4 weeks.1 ,68 Long-term sequelae following exposure to these agents are rare.34 ,42
Management
The optimum treatment for individuals affected by OC, CS or CN is currently based on results from case reports or case series as high-level evidence for the best treatment is not available. There is no antidote for these agents with treatment mainly consisting of thorough decontamination and symptom-directed supportive care.
Removal of those exposed from the contaminated area and into fresh air is the most important initial undertaking.83 Aerosolised tear gases are heavier than air, and any exposed patient who has lost consciousness or is lying should be lifted off the ground; emergency response vehicles should also try and park in higher areas.2 Transport to a medical facility is recommended for symptomatic exposures. A concern with the medical management of those affected by CN or CS is secondary contamination of first responders such as police or ambulance, or the medical staff at the hospital. While it is uncommon for there to be heavy contamination of people following exposure to these products, there is a risk of secondary exposure occurring. For example, among attending physicians treating exposed patients, minor effects such as facial pruritis and respiratory and eye irritation may develop.34 ,37 ,40 ,84 Removal of contaminated clothing, prompt decontamination, the use of gloves, goggles, gowns and surgical masks by medical personnel, and, if possible, treatment in a well-ventilated room are recommended to minimise secondary contamination.2 ,33 ,40 ,61 ,83 ,84 Removed contaminated clothing should be sealed in a double plastic bag.12
Treatment for ocular exposures initially requires thorough eye decontamination. Flushing the eyes with water or saline for 10–20 min is the most often recommended initial treatment for decontamination of the eyes.1 ,2 ,27 ,33 ,34 ,61 ,85 Some have recommended using air flow over the eyes, such as that directed from fans or the use of a cold hairdryer to decontaminate the eyes following exposure to aerosolised smoke products.18 ,48 ,68 ,86 ,87 This is purported to help the gas vaporise and therefore relieve irritation.85 However, it seems improbable that a powder that dissolves into solution on the surface of the eye would readily be converted into a gas at normal temperature and pressure even with air flow over the eyes.33 This technique is also unlikely to be effective for liquid solvent spray products.88 Additionally, some studies have noted that cold air blowing over the face and eyes did not produce any clinical improvement and only served to increase contamination of the treatment facility, whereas flushing with normal saline resulted in considerable improvement in symptoms.33 Based on this review of the literature, flushing with saline or water for 15–20 min would appear to be the most sensible initial procedure for any exposure to CN or CS. Patents may require a topical anaesthetic to enable them to open their eyelids sufficiently for effective irrigation. Contact lenses should be removed before flushing.
If following irrigation more than mild, resolving symptoms are present, a full ophthalmological examination should be undertaken, including fluorescein staining and slit-lamp examination. For persistent symptoms or if there is injury noted, then specialist ophthalmological assessment is recommended.1 ,33 ,85 Further treatment may need to include oral analgesics, topical antibiotics and a cycloplegic or mydriatic.1 ,85 In some situations, for example, when a tear gas grenade explodes in close proximity to the face, there may be particles embedded in the cornea or conjuctiva.49 Following flushing, removal of these particles is necessary; use of a cotton wool swab or a needle tip at a slit lamp is recommended.1 ,49 ,89
Skin
Skin exposures should be thoroughly decontaminated with copious flowing water and soap to remove the contaminate and settle the burning sensation.80 The face should be wiped to remove any particles before being washed.33 ,34 ,39 ,61 ,83 ,85 Saline irrigation should be used for vesiculated skin.1 Other decontamination methods have been investigated; a mild alkaline solution (sodium carbonate or sodium bicarbonate) or a sodium metabisulfite solution has been recommended,6 ,25 ,31 ,90 although these may not always be available and are unlikely to provide much benefit over water,31 thus they are probably unnecessary. Baby shampoo was found to be no better than water alone in one randomised trial.91 Diphoterine, a commercial decontamination solution purportedly containing chelating and amphoteric properties, has been investigated with some positive findings,92 as has the use of vegetable oil88 though further evidence is required before advocating their use. It is rare that severe contact dermatitis occurs,87 in which case it is generally treated with topical corticosteroids and/or antihistamines such as diphenhydramine; systemic steroids may be required if symptoms are severe. Significant chemical burns should be treated in the same way as thermal burns.1 ,18 ,33 ,41 ,43 ,64 ,81 ,83 ,87
Respiratory
While the majority of respiratory symptoms are mild and should improve with cessation of exposure and removal to fresh air, high concentrations (such as exposure in a confined space) or prolonged exposure periods may cause significant respiratory symptoms.24 Monitoring and support of respiratory function is important in any symptomatic patient. Pulse oximetry and arterial blood gases should be monitored. If significant respiratory distress develops, initial supportive treatment includes oxygen administration.33 ,55 ,61 ,62 Chest radiographs can assist in identifying any pulmonary complications.4 ,61 Suctioning may be required for those with copious respiratory secretions.56 Along with continued oxygen therapy, inhaled bronchodilators such as β-2-agonists (ie, salbutamol) may assist those with bronchospasm and/or obstructive changes on lung function tests.34 ,38 ,39 ,64 Inhaled steroids may also assist in patients with bronchospasm (or non-productive cough).38 Respiratory failure may rarely occur secondary to laryngospasm; airway protection and assisted ventilation may be required.37 ,61 ,62
Exacerbation of asthma, emphysema or bronchitis may occur in those with a pre-existing respiratory condition, or uncommonly, asthma may develop due to an allergic respiratory response to the agent.6 ,31 ,33 ,42 ,55 ,64 ,93 Standard asthma treatment protocols should be followed. Reactive airways dysfunction or pulmonary oedema has only rarely been reported and may be delayed or exercise induced.4 ,61 Pulmonary oedema is managed primarily by non-pharmacological treatments, including resting from activity and oxygen therapy. Standard face mask supply of 50–60% oxygen may maintain adequate oxygenation. However, mechanical ventilation with continuous positive airway pressure or positive-end-expiratory pressure ventilation may be required if PaO2 cannot be maintained above 50–55 mm Hg.
Gastrointestinal
While gastrointestinal symptoms are not common, retching, nausea and vomiting can occur due to the irritant effects.39 ,55 ,94 Some people appear to be especially sensitive to the effects and may vomit more readily, while vomiting also appears more common when high concentrations are attained, such as when exposure occurs in a confined space or when a long duration of exposure occurs.55 Ingestion of contaminated saliva may also contribute to vomiting and diarrhoea. Very rarely actual ingestion has occurred and led to gastrointestinal disturbances.1 ,39 There are limited data on the benefit or otherwise of gastrointestinal decontamination procedures following ingestion. Due to the relatively minor effects expected following ingestion, the risk of adverse effects from decontamination likely outweighs any potential benefit. Gastrointestinal decontamination with gastric lavage or activated charcoal is therefore not recommended. Overall gastrointestinal symptoms typically resolve spontaneously, and further specific treatment is not required.1 ,32 ,39 However, if vomiting or diarrhoea is persistent or severe, this may contribute to fluid and electrolyte imbalances, acidosis, shock, seizures, obtundation and hypokalaemia.95 In this situation, patients may require symptomatic care with intravenous rehydration, antiemetic agents and adequate electrolyte replacement.
Conclusion
CS, OC and CN are effective lacrimating agents and are therefore ideal for riot control. Significant clinical effects are not expected following the majority of exposures. Beyond their capacities to cause eye and skin irritations, symptoms can also include photophobia, conjunctivitis and periorbital oedema, laryngospasm and bulla formation and subcutaneous oedema. These can be minimised both by rapid evacuation from sites of exposure and appropriate supportive care of the intoxicated patient. Treatment is largely symptomatic and supportive with emphasis on decontamination, monitoring and support of respiratory function.
References
Footnotes
-
Contributors LJS preformed initial literature review, drafted the initial manuscript, and approved the final manuscript as submitted. RJS assisted with literature review, contributed to initial draft, reviewed and revised the manuscript, and approved the final manuscript as submitted. DIMcB contributed, reviewed and revised the manuscript and approved the final manuscript as submitted.
-
Competing interests None.
-
Provenance and peer review Not commissioned; externally peer reviewed.